Research overview
Bacteria have the capacity to efficiently adapt to environmental changes by sensing the availability of nutrients. Because glucose is one of the most abundant carbon sources in the environment, it is the most favored carbohydrate in most organisms. Although different organisms employ different uptake systems for glucose, many organisms, including yeasts and E. coli, show preferential growth on glucose when presented with mixtures of glucose and other carbon sources. It is known that glucose, sensed by its transporters or metabolizing enzymes, inhibits the uptake of certain other sugars into the cell or represses the synthesis of enzymes required to grow on those alternative carbon sources such as lactose.
In most bacteria, glucose and many other carbohydrates are transported via the phosphoenolpyruvate (PEP):sugar phosphotransferase system (PTS). This multi-component system carries out a PEP-dependent transport and phosphorylation of numerous sugars (group translocation; Fig. 1). There is a sequential phosphoryl transfer to two general proteins, enzyme I (EI) and HPr, then to membrane-associated enzymes II, which may consist of 3–4 domains (A, B, C, and D). Over the years, this system has become established as a central component in bacterial physiology as a result of the identification of multiple regulatory functions, superimposed on the transport functions, for essentially all the components of the glucose arm of the PTS (Fig. 2).
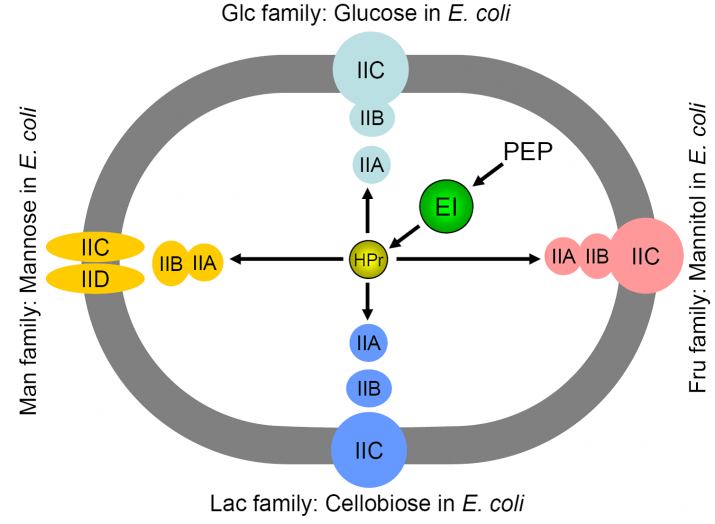
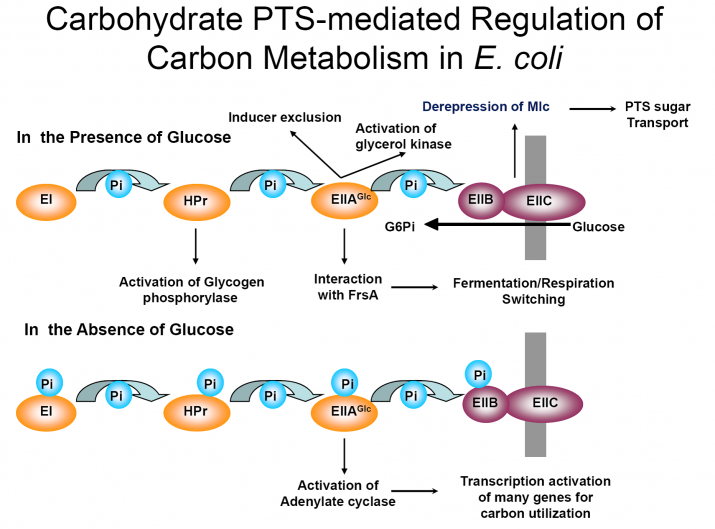
Fig. 1. PEP:sugar phosphotransferase systems in E. coli. Fig. 2. PTS-mediated regulation of carbon metabolism in E. coli.
Analysis of the Escherichia coli genome has revealed new phosphotransferase-encoding genes. The novel nitrogen-metabolic phosphotransferase system (PTSNtr) proteins, NPr and EIIANtr (paralogues of HPr and EIIAFru, respectively), are encoded by the ptsO and ptsN genes, respectively, localized to the rpoN operon of E. coli. Enzyme INtr (EINtr; enzyme I paralogue) encoded by the ptsP gene, located distant from the rpoN cluster on the chromosome, consists of two domains, an N-terminal domain of 127 amino acids homologous to the N-terminal sensory domain of the NifA protein of Azotobacter vinelandii and a C-terminal domain of 578 amino acids homologous to all currently sequenced enzymes I. It was shown that the E. coli nitrogen-metabolic PTS consists of the following phosphoryl transfer chain, PEP→EINtr→NPr→EIIANtr (Fig. 3).
Because phosphoryl transfer to a specific substrate has not yet been demonstrated for the PTSNtr, it has been suggested that the role of this pathway is in regulation. In contrast to the extensive body of data dealing with regulation mechanisms associated with the carbohydrate PTS, substantially less is known about regulatory mechanisms connected with the nitrogen-metabolic PTS.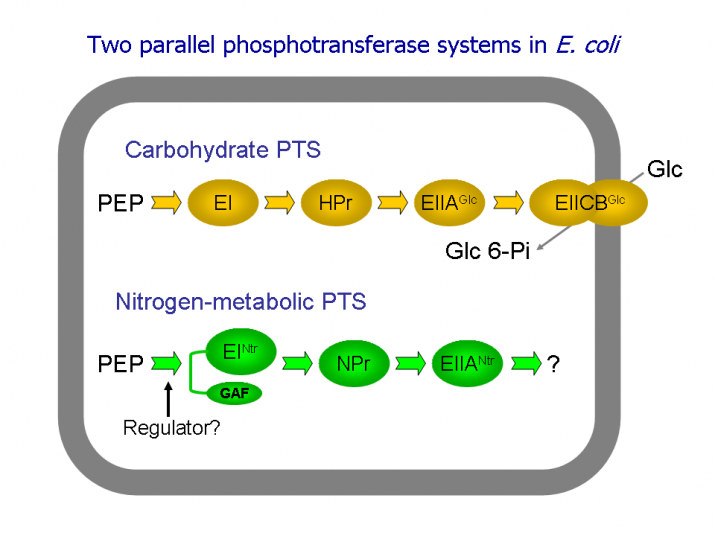
Fig.3. Two parallel PEP-dependent phosphotransferase systems in E. coli.
What we are currently working on:
- The mechanism how the PTS is related to bacterial pathogenicity.
- The mechanism how bacteria sense the absence of amino acids.
- The mechanism how the intracellular potassium level regulates gene expression.
- The exact mechanism for the regulation of adenylate cyclase activity by IIAGlc.
- Regulatory roles of NPr in stress responses.
- Role of the GAF domain in EINtr.
