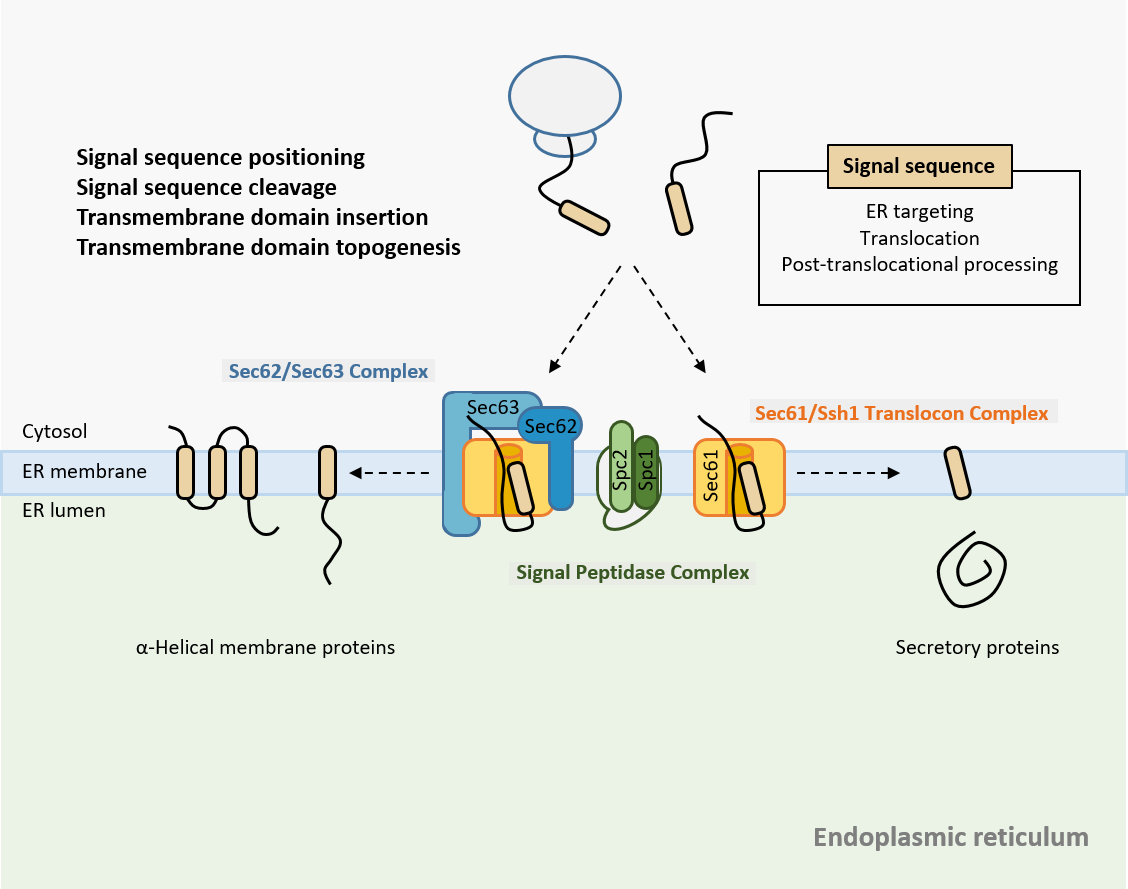
Membrane proteins make up nearly 30% of the cellular proteome and play vital roles in cell-to-cell communication and the transport of molecules across membranes. The proper function of these proteins relies on a finely tuned balance of synthesis, quality control, and degradation—a process collectively known as membrane proteostasis. When this balance is disrupted, it can lead to protein misfolding or overexpression, contributing to the development of various diseases.
Despite their biological and clinical significance, the mechanisms that govern membrane proteostasis remain largely elusive. Our lab is dedicated to uncovering the fundamental principles that cells use to maintain the health of the membrane proteome.